Nalidixic Acid Powder
Product Details:
- EINECS No 206-820-0
- Boiling point 300 C.
- Storage Room Temperature
- Molecular Weight 232.24 Kilograms (kg)
- Melting Point 232 234 C (450 453 F)
- Particle Size <100 microns (fine powder)
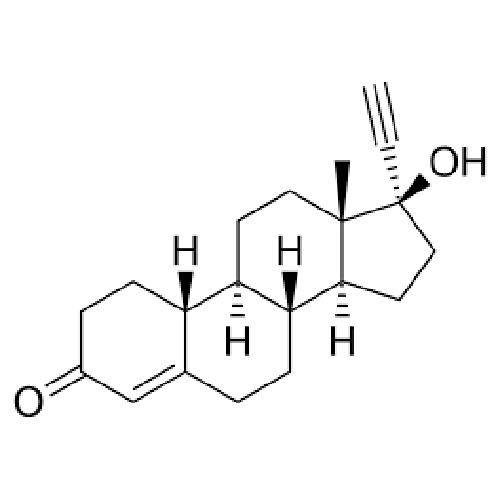
- Structural Formula O || N C C C C C C C C C C / | \ C N C | | CH3 COOH
- Click to View more
Nalidixic Acid Powder Price And Quantity
- 25 Kilograms
- 4800.0 INR/Kilograms
Nalidixic Acid Powder Product Specifications
- Powder
- 206-820-0
- Room Temperature
- 300 C.
- Medicine Grade
- 1-ethyl-7-methyl-4-oxo-1,8-naphthyridine-3-carboxylic acid
- 232.24 Kilograms (kg)
- 100%
- 232 234 C (450 453 F)
- Pharmaceutical Intermediates
- NegGram
- <100 microns (fine powder)
- O || N C C C C C C C C C C / | \ C N C | | CH3 COOH
- 2933.99
- 4.5 to 5.0.
- 20ppm
- Nalidixic acid is primarily used as an antibacterial agent, particularly effective against Gram-negative bacteria. It was the first quinolone antibiotic, and while it has been largely replaced by newer quinolones like ciprofloxacin, it is still used in certain contexts.
- No Smell
- white to light yellow crystalline powder
- approximately 50 mg/ml
- Not more than 0.5% to 1.0%
- 389-08-2
- 12 Months
- C H N O
Nalidixic Acid Powder Trade Information
- Mumbai Port
- Cash Against Delivery (CAD), Cash Advance (CA), Cash in Advance (CID), Days after Acceptance (DA), Letter of Credit at Sight (Sight L/C), Telegraphic Transfer (T/T), Letter of Credit (L/C)
- 100 Kilograms Per Day
- 7 Days
- No
- Free samples are available
- DRUM PACKING
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- WE PROVIDES ALL KIND OF CERTIFICATIONS AS YOU REQUIRED
Product Description
In terms of bacteriostatic and bactericidal effects, nalidixic acid depends on the microorganisms sensitivity and the concentration. In addition, it has demonstrated efficacy against Gram-negative bacteria, such as colon bacillus, shigella, proteus, salmonella, and Fridlenders bacillus.
Properties:
- Formula: C12H12N2O3
- CAS ID: 389-08-2
- ATC code: J01MB02 (WHO)
- Elimination half-life: 6-7 hours, significantly longer in renal impairment
- Molar mass: 232.239 gmol
- Synonyms:1,4-Dihydro-1-ethyl-7-methyl-1,8-naphthyridin-4-one-3-carboxylic acid, 1-Ethyl-1,4-dihydro-7-methyl-4-oxo-1,8-naphthyridine-3-carboxylic acid
- CAS Number:389-08-2
- Molecular Weight:232.24
- Density:1.33 g/cm3
- Boiling Point:413.1 C
- Melting Point:227-229 C
- Form:Powder
- Color:White to light yellow
How does it work?
Nalidixic acid is known as a quinolone antibiotic that interferes with a bacterial enzyme that reproduces and repairs the genetic material (DNA) of bacteria. The enzyme is essential for bacterial reproduction and repair.
Applications Or where it is used:
This medication treats urinary tract infections caused by susceptible gram-negative microorganisms, such as Klebsiella species, E. Coli, Enterobacter species, and Proteus species. A quinolone antibacterial, Nalidixic Acid is taken orally.
Dosage of usage:
Adults
Adults are prescribed a dosage of 1 g administered four times daily for a period of one or two weeks (3 g daily total). After the initial treatment period, the daily dose may be reduced to 2 g. If the dosage is too low during the initial treatment, bacterial resistance may develop.
Pediatric Patients
The use of Nalidixic Acid on infants under three months of age should be avoided until further experience is gained. Children aged 12 and under should be dosed based on body weight. To initiate treatment, the daily dosage should be 25 mg/lb (55 mg/kg) divided into four equally spaced doses. If treatment is prolonged, the dose can be reduced to 15 mg/lb/day (33 mg/kg/day). The suspension or capsules of 250 mg may be used. Each 250 mg capsule equates to a teaspoon (5mL) of suspension.
Side effects:
Nalidixic Acid has been linked to the following reactions after oral administration:
- CNS effects
- Drowsiness
- Weakness
- Headache
- Dizziness
- Vertigo
There have been very few reports of reversible subjective visual disturbances without objective evidence (generally occurring with each dose during the first few days of treatment). A wide range of reactions can occur, including over-bright lights, altered color perception, difficulty focusing, a loss of visual acuity, and double vision. These effects usually disappeared when the dosage was reduced or the therapy discontinued. Occasionally, there have been cases of toxic psychosis or brief convulsions following excessive doses. Patients with epilepsy or cerebral arteriosclerosis are generally more prone to convulsions. The treatment of newborns and children with nalidixic acid may occasionally result in increased intracranial pressure with bulging anterior fontanels, papilledema, and headache. Sixth cranial nerve palsy has been reported in some cases.
Patients with CNS disorders, such as cerebral arteriosclerosis or epilepsy, or those who are susceptible to seizures should use Nalidixic acid with caution. Nalidixic acid should not be administered to patients exhibiting these adverse reactions.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+













 : nilesh.sheth70
: nilesh.sheth70
